Nouvelles

A termite mound on the road to Tom Price, Western Australian. A 2014 field trip through the Pilbara with two of the author’s PhD students, Jake Coates and Holly Caravan (not the first trip to Australia for any of them)
A Canadian Entomologist in Australia (it has been done before, I am sure, but here are my two cents. Or, should I round that down to zero or up to 5 cents?) Okay, new title: My five cents
Guest post by Tom Chapman
I have introduced many Canadian students to Australian based fieldwork (e.g. Jake Coates). They’ve heard the stories, so they tended to start with some fear of Australia’s deadly denizens. Here is my advice to them woven into some of my personal stories of working in Australia. First, some of the most amazing biologists (professional and otherwise) in the world are Australian. But, do not assume that every Australian you meet is an outback survival expert with excellent knowledge of the local flora and fauna. And get ready; you are going to be teased and fed a lot of nonsense (exhibit A: dropbears).
My first expedition to Australia, the land of perilous animals, was in 1997. I was a graduate student, I knew next to nothing about the southern hemisphere let alone anything about Australia, and I was traveling on my own. I was to begin collecting gall-inducing thrips on Acacia. My supervisor, arguably the world’s expert on these insects (sorry Laurence Mound, father of all things thrippy), was to follow me a few weeks later. Why was I going first? I never really knew, maybe my supervisor didn’t want to be seated long-term by my side on cramped airplanes, but I tried not to take it personally. After about 31 hours of traveling, I arrived in Adelaide; well placed at the edge of Australia’s arid zone to begin my search for Acacia thrips.
A very generous and outgoing student, among the research group at Flinders University that was to host me, volunteered to pick me up at the airport. It was a two hour round trip for her, so I was grateful and indebted, but she seemed to have no idea how exhausting my travels were for me. During our commute to the University she was non-stop questions and instantly personal. What were my dreams and aspirations? Did I prefer to sleep with men or women? How many times had I had my heart broken? My brain was so clouded with exhaustion that I couldn’t deflect this assault or form coherent replies, the latter of which didn’t seem to matter to her. When we parked at Flinders, we were confronted by a long flight of cement stairs leading to the biology building. I lagged well behind on our ascent, but here is when I uttered my first ignorant and anxiety-motivated question during this visit: Should we be worried about redback spiders? She came back down the stairs and told me that while having lived her entire life in Adelaide she had never seen a living specimen of that species. I pointed down and past her foot and I said, “I think that’s one”. She took a look and she agreed: it was a living redback spider. We continued up the stairs, but I was baffled. How had this woman, so unaware of this deadly spider species, survived to adulthood? Anyway, one hour after arriving in Australia I had escaped injury during this my first deadly Australian animal encounter.
Over the next few days my jet lag receded and I started to get to know many more of the students among my host group. I knew I was making strong personal connections when during a trip to the campus bar one student informed me, “We thought you’d be an asshole”. After asking a few questions, what I think he was saying was that having met my supervisor the year before, that by association I would share many of his qualities. I don’t agree with this person’s opinion of my supervisor, but is there evidence of personality associations between students and supervisors? I leave you, the reader, to ponder that question, and I know that for some of you it would be horrifying if there were positive evidence on the subject. Another indication that I was making connections that afternoon was that I was also invited to join a group to watch a Cricket test on TV. I didn’t admit it at the time, but I thought Cricket was a game that died out a century and a half ago. I can’t have been the only Canadian that is embarrassingly ignorant of the fact that there are well over a billion people that are obsessed with this game. And for those that are aware of the vibrancy of Cricket, and think very little of me now, I want you to know that I became a fan. For instance, I know who Sir Donald Bradman is and I even lived a few blocks away from Centennial Park Cemetery and was present when his ashes were interred there. If you want to impress South Australians in particular, look up “The Don” and memorize a few of his batting statistics, you’ll win over some hearts.
However, this initial introduction to Cricket was painfully dull for me despite my host’s encyclopedic tutorials on rules and traditions. Several times I tried to engage the group in conversational topics other than Cricket. These efforts failed until I asked about swimming locally and the potential for being attacked by sharks. Admitting any anxiety about these dead-eyed predators to a group of Australians is somewhat like the popular notion of adding blood to the water on the behaviour of a shark. Everyone in that lounge room broiled with horrendous attack stories for me to hear. It was hard to keep track, but I think there were at least three people this group knew of directly who had been bitten or killed. When they had clearly shaken me the group switched to trying to assure me that swimming was safe – Listen mate, you have more chance of being struck by lightning. I asked the group to tell me some stories of people they knew that had been struck by lightning. They didn’t have a one! I am not suggesting that means that shark attacks are more common; instead I think it means that lightning strike stories just don’t hold the attention of visitors to Australia. Therefore, there isn’t the same temptation for locals to retell, confuse some details and exaggerate these stories. I have seen other visitors tormented the same way as I was. It seems cruel. We really are worried and have deep fears about shark attacks. Why is that not apparent to our hosts? I think I gained some insight years later when I moved to Adelaide and naturalized (my family and I became CanAussies). A neighbour asked me how, when I lived in Canada, I had the courage to leave my house. I thought they were referring to Canada’s cold winter weather, but instead they meant the bears. Doesn’t that sound ridiculous? Even Canadians that live in bear country would find that ridiculous – Listen mate, you have more chance of being struck by lightning (I’m strategically leaving Churchill, Manitoba out of this discussion). My point is that Australians see our fears as absurd so teasing us doesn’t seem so wrong.
Australians might have sharks in perspective, but not everyone you meet there knows the bush like a Mick Dundee. Australia is more urbanized then Canada (89.2 % versus 80.7 %). And, among the general population there remains significant fear and ignorance of the wildlife on their Island Continent. Turning again to the deadly redback spider, another neighbour in Australia was using these nifty rake/gloves to bag yard waste. Imagine The Wolverine with webbing between his claws. A redback climbed out of the dried leaves pinned between the gloves and crawled on to the back of my neighbour’s hand and bit him. He told me later that the pain was immediately blazing hot and he was terrified that he would die. That is not what is commonly reported; the bite is usually described as a mild sting with pain sharpening 20 to 40 minutes later. He screamed, got the attention of his wife and she rushed him to emergency where the highly competent staff there encouraged him to ice it, monitored him for a little while and then sent him home. What, not instant death? There is an antivenin but it is not always administered, and there hasn’t been a death due directly to a redback bite post 1956 (when the antivenin was developed). We found a redback in the pouch on the front of my daughter’s bicycle, one in the door of our car, and one under the last step of the spiral staircase in our house. I played volleyball once a week at a sports complex and if our game was the last of the evening we had the job of taking down the nets and turning off the lights. One night a teammate noticed that there was a redback in the light box. He warned us that they can jump two meters and that we needed to stay back. Nonsense. Enough was enough. I stepped up and reached in and turned off the lights. I am certain that this spider was grateful; the dark brings out her preferred prey and it certainly wasn’t volleyball players. While living in Australia, these spiders were a constant in my family’s lives and none of us were ever bitten. It was now easy to imagine how the unobservant woman I mentioned at the start of this piece survived her childhood and adolescent years.
It is now almost two decades that I have been conducting fieldwork in arid Australia. The only animals that have caused me any harm have been ants, it was on that first trip to Australia and it wasn’t that bad. By this time my supervisor had arrived in Australia. Along with an Australian student, my supervisor and I drove from Adelaide to near Brisbane and returned to Adelaide. The trip took us 10 days and we covered over 5000 km, much of it on dirt tracks. We kept the air conditioner off to save fuel and we had the windows down. The work was hot and dusty. I wanted to be seen as a hard working student. I didn’t want to show any weakness on this trip, but by the sixth day late in the afternoon a wall appeared and I ran right into it; I had squatted down in front of a small Acacia bush and was staring blankly through the foliage. I hoped that it would appear that I was still looking for thrips galls, but I was really pretending that I was anywhere but in that desert. So I didn’t notice that several hundred bull ants had crawled up over my boots and socks until they started stinging me. I whooped and leapt around while slapping myself with my hat, and just as I settled down a flat bed pickup truck, off road and appearing to come from nowhere, drove slowly (a trot maybe) past me. There were four people in the cab, two on the cab, two on the bonnet and maybe eight people on the back. In the middle of the eight was a very large and dead red kangaroo: a big boomer. Even though the truck was only meters away, no one made eye contact with me as they past except for a little kid that beamed me a beautiful smile and waved the Kangaroo’s front paw. All these years later that moment remains my most favorite, stings and all.
We took a family vacation to Alice Springs in the Northern Territory. It was a three-day drive up from Adelaide, tough to do with little kids. We had only two children’s music tapes, so we heard the tapes a dozen times each. One tape was by the Wiggles. It was the one where the Wiggles ask Steve Irwin (the Crocodile Hunter) a question about an Australian animal, like can emus fly? After Steve answered them the Wiggles would then sing a song about that animal. After we heard this tape five times my five-year-old sounding very exasperated bellowed, “Those Wiggles don’t know anything about Australian animals!” To be fair, they probably do know something, and I would say that just like hiking through Canada’s bear country it helps to have a little knowledge about the local wildlife to stay safe. But, fear and ignorance have no place. While traveling in Australia, if you still get talked into putting forks in your hair to ward off dropbear attacks, well then there is no helping you.
This past weekend marked the beginning of the Entomological Society of Canada and the Société d’entomologie du Québec’s Joint Annual Meeting in Montreal. This three day event brought together a large number of insect researchers and insect enthusiasts from all across Canada. This was my second ESC/SEQ meeting in Montreal, and the second since I have been a student. As a blog administrator, I got a bit of an inside look at the current issues facing the society at the meeting of the ESC board meeting, which will be the subject of future posts. I also got quite a few bedbug bites from staying in a cheap hostel the night of the board meeting, but that is another, and terrible story.
Anyway, of course I brought my camera, and so here I give you the conference from my perspective.

On the opening day, the Gold Medal address was delivered by Jon Sweeney, reflecting mainly on his collaborators over the years and how the have helped shape his stellar career in entomology.

Guy Boivin delivered the Heritage Lecture, which was an awesome mix of First Nations insect lore, followed by the early natural historians of New France. I learned quite a bit from this, and I hope Guy may write some more on the subject for the Canadian Entomologist.

Sunday’s plenary session featured Marcel Dicke from Wageningen University, and was an absolutely fascinating story about herbivores, parasitoids and hyperparistitoids on mustards. The interactions he described kind of blew my mind.

The first talk of the Graduate Student’s Showcase was by Christina Hodson from UVic. She described her work on a charismatic little psocopteran and its weird sex distorting elements.

Holly Caravan of Memorial University delivering her lecture on fascinating social aphids, with some great background on other social insects.

Jean-Philippe Parent of Université de. Montréal gave a riveting lecture on how to determine if an insect can measure time.

Leanna Lachowsky of University of Calgary with a topic near and dear to those of of from the west: mountain pine beetle! This was a cool study on sex allocation in this troublesome forest pest.

After the great opening sessions, we all repaired to the Insectarium to enjoy drinks in the company of our favourite colleagues and study subjects!

If you ever try photographing people in this space, you will quickly learn how much colour casts arise from the brightly painted walls. I did manage to capture this one of Louise as many of you will remember her, behind the camera!

Big thanks to Sarah Loboda and Maxime Larivée for running so much behind the scenes. They provided to me my favourite shot of the conference as well! Not sure how they kept their wits about them, but I think it was because they both have such a good sense of humour.

Monday’s plenary was delivered by Jessica Forrest, from University of Ottawa, talking about a whole range of issues with a population of montane bees in Colorado.
From here on, my trajectory through the conference will probably differ substantially from yours. I of course needed to attend the sessions in which my former labmates were giving talks, but even so I did not manage to catch them all! I present to you instead a slideshow of images that I took during the conference. I will say how impressed I was by the student presentations this year in the GSS and the President’s Prize sessions. ESC students are really on the ball at how to give effective talks, and I hope that the more senior among us are paying attention! Perhaps in 2017 we can have a Student’s Prize to award to the best regular session talk!
-

- Memorial in the J.B. Wallis and R.E. Roughley Museum of Entomology with a case of Dr. Roughley’s Dyticid beetles.
-
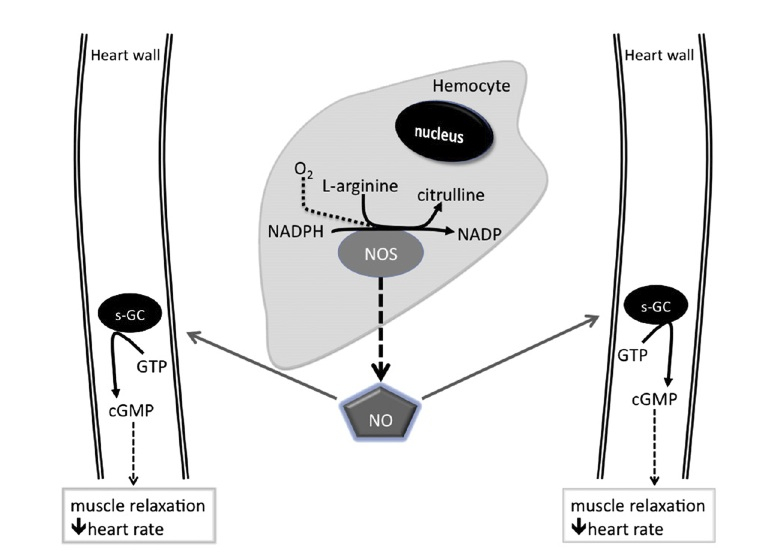
- Schematic diagram of the proposed regulation of cardiac activity in B. extradentatum by the gaseous signaling molecule, nitric oxide (NO). Figure 7 from da Silva et al. 2012.
-

- View from my parents’ house – on clear days you can see all the way to Lake Ontario. Photo: Kathleen Timms
-

- A carpenter bee, Xylocopa virginica, sitting on a Weigela flower and taking nectar through a hole it has cut in the base of the corolla. Photo: Laura Timms
-

- Galls on a red oak, Quercus rubra, tree. Most are at the base of a branch. Some of the galls have had lots of adults emerge (note the emergence holes), and some have not. Photo: Laura Timms
As part of a continuing series of Canadian Entomology Research Roundups, here’s what some Canadian entomology grad students have been up to lately:
Ecology and Evolution
Rasoul Bahreini (University of Manitoba) found that honeybee breeding can improve tolerance to Varroa mites which can help minimize colony losses in the winter and improve overwintering performance (Article link). Rasoul also found that reducing ventilation may be an effective way to manage Varroa mite infestation in overwintering honeybee colonies (Article link), and that Nosema infection restrained Varroa removal success in bees (Article link).
A novel method based on agar-polydimethylsiloxane devices to quantitatively investigate oviposition behaviour in Drosophila melanogaster was described by Jacob Leung and colleagues (York University) (Article link).
Paul Abram (Université de Montréal) and his colleagues found that a predatory stink bug has control of egg colouration, depending on whether it is laying on the top or underside of leaves. The pigment protects developing embryos against UV radiation (Article link). See also a related post on the ESC blog, an article in the New York Times, and a dispatch article in Current Biology.

A spined soldier bug (Podisus maculiventris) female, with the range of egg colours she is capable of laying (Photo: Leslie Abram/Paul Abram/Eric Guerra)
Philippe Boucher and colleagues (Université du Québec à Rimouski/Chicoutimi) found that ant colonization of dead wood plays a role in nitrogen and carbon dynamics after forest fires (Article link).
Did you know that ground squirrels have lice – and males have more than females? Neither did we, but Matt Yunick and colleagues (University of Manitoba) recently published an article in The Canadian Entomologist describing their findings (Article link).
Boyd Mori and Dana Sjostrom (University of Alberta) were part of a group of researchers that found that pheromone traps are less effective at high densities of forest tent caterpillars because of competition for pheromone plumes (Article link).
Parasitoid memory dynamics are affected by realistic temperature stress. As part of a collaboration with the University of Palermo (Italy), Paul Abram (Université de Montréal) and colleagues discovered that both hot and cool temperature cycles prevent wasps (Trissolcus basalis) from forgetting. (Article link).

Trissolcus basalis (Hymenoptera: Platygastridae) wasps (left panel) parasitizing the eggs of their host stink bug Nezara viridula (Hemiptera: Pentatomidae; mating couple shown in right panel). These parasitoids can detect their host’s « chemical footprints », and even commit them to memory! (Photos: Antonino Cusumano)
Crisia Tabacaru and Sarah McPike (University of Alberta) studied Dendroctonus ponderosae and other bark and ambrosia beetles and found that competition between the beetles may limit post-fire colonization of burned forest stands (Article link).
Marla Schwarzfeld (University of Alberta) found that tree-based (GMYC and PTP) species delimitation models were less reliable in delimiting test species, and the Nearctic Ophion (Hymenoptera: Ichneumonidae) fauna is much larger than previously thought (Article link).
Where have all the mosquitoes gone? Emily Acheson and colleagues (University of Ottawa) found spatial modelling reveals mosquito net distributions across Tanzania do not target optimal Anopheles mosquito habitats (Article link).
Tyler Wist and colleagues (University of Alberta) found that a native braconid parasitoid (Apanteles polychrosidis) uses host location cues induced by feeding damage on black ash but not on green ash (Article link). Also check out the author’s recent post on the ESC Blog!
Agriculture
Sharavari Kulkarni and colleagues (University of Alberta) discovered that reducing tillage could increase the amount of weed seeds consumed by carabid beetles (Article link).
Physiology and Genetics
Sebastien Boutin and colleagues (Université Laval) are beginning to decode the genetic basis of honeybee hygenic behaviour (Article link).
Investigating the cold tolerance of different Sierra leaf beetle life stages, Evelyn Boychuk and colleagues (University of Western Ontario) found that adults are freeze tolerant, the eggs and pupae are freeze-avoidant, and the larvae are chill susceptible (Article link).
From the Authors:
Shaun Turney, Elyssa Cameron, and Chris Cloutier had this to say about their new article published in PeerJ:
Our supervisor, Prof. Chris Buddle, has always emphasized the importance of voucher specimens for our entomology research. He explained that voucher specimens make our work replicable and verifiable. We wondered how widespread the practice of making voucher specimens among those practicing arthropod-based research. We investigated the frequency of voucher deposition in 281 papers, and the factors which correlated to this frequency. Surprisingly, vouchers were deposited less than 25% of the time! Our paper highlights the need for a greater culture of voucher deposition and we suggest ways in which this culture can be cultivated by researchers, editors, and funding bodies.

Voucher specimens: an important component of arthropod-based research (Photo provided by Shaun Turney, Elyssa Cameron, and Chris Cloutier)
From Ikkei Shikano, on two of his recently published articles:
Parents that experience a stressful environment can equip their offspring to fare better in a similar environment. Since this can be energetically expensive for the parent, we asked if parents are exposed to two stressors (nutritional stress and a pathogen), would they equip the offspring for both stressors or would they select one over the other? Cabbage looper moths exposed to a pathogen and poor food quality produced offspring that were highly resistant to that same pathogen. Parents that were given poor food produced offspring that developed faster on poor food. When the parents experienced both stressors, they produced offspring that were resistant to multiple pathogens but did not grow faster on a poor diet (Article link).
Herbivorous insects unavoidably eat large and diverse communities of non-entomopathogenic microbes, which live on the surface of their host plants. Previous studies suggest that consuming non-entomopathogenic bacteria may induce a costly immune response that might decrease the risk of infection by pathogens. But isn’t it wasteful for an insect upregulate a costly immune response to non-pathogens that it ingests with every meal? Within an appropriate ecological context, we show that cabbage looper, Trichoplusia ni, larvae do not induce a costly immune response, indicating that they are adapted to consuming non-pathogenic bacteria that are commonly found on the surface of their host plants (Article link).
From Kate Pare, on an article published by a group of undergraduates taking the Arctic Ecology field course at the University of Guelph:
Our study focused on changes in ant diversity in the area surrounding Churchill, Manitoba between the historic collections made by Robert E. Gregg in 1969 and collections made by students and instructors of the Arctic ecology field course in 2012. Seven ant species were collected in 2012 compared to the five species recorded from 1969. This increase in species richness in the 2012 collection is more likely a result of cryptic molecular diversity that was overlooked in the collection made in 1969 (Article Link, post on the ESC blog).
The ESC Student Affairs Committee will be continuing to help publicize graduate student publications to the wider entomological community through our Research Roundup. If you published an article recently and would like it featured, e-mail us at entsoccan.students@gmail.com.
For regular updates on new Canadian entomological research, you can join the ESC Students Facebook page or follow us on Twitter @esc_students.
By Tyler Wist
The ash leaf cone roller, Caloptilia fraxinella (Ely) (Lepidoptera: Gracillaridae) (Fig. 1) started to get noticed in the cities of the Western Canadian prairies in 1998, well, in Saskatoon, SK at least. I know this because that summer the green ash, Fraxinus pennsylvanica (Oleaceae), in my front yard was covered in cone rolled leaflets and had not been prior to that year. I had just started working for the City of Saskatoon’s Pest Management Program that year and one of our mandates was urban forest insects…not that there was any budget to control them, but it piqued my interest in urban forest entomology.

Fig. 1 – The ash leaf coneroller, Caloptilia fraxinella (Ely) (Lepidoptera: Gracillaridae) adult, pupal exuvium and cocoon.
The following year, Chris Saunders with the City of Edmonton’s Pest Management Program, contacted us in Pest Management and asked if we had seen this cone roller on our ash trees because they had just noticed it on the ash trees in Edmonton. Greg Pohl had identified this leaf miner/leaf roller that year on all species of horticultural Fraxinus in Edmonton and published the identification and some life history of the moth in a 2004 paper (Pohl et al. 2004) along with a brief identification of several parasitoids that were reared from larvae and pupae. The lone braconid, identified to the genus Apanteles and found to be all one species by Darryl Williams of the Canadian Forest Service in Edmonton seemed to be the dominant parasitoid in this complex, but without a species designation not much else about the wasp could be gleaned from the literature.
Chris Saunders suggested that I study the ash leaf cone roller as a master’s project but I digressed from urban forest entomology for a few years into pollination of a nutraceutical/agricultural crop. By this time, the ash leaf cone roller had spread to every ash tree in both cities and often rolled 100% of the leaflets on a single tree. I finally followed Chris’ advice and started a PhD project in Maya Evenden’s lab at the University of Alberta, which was the only lab in Canada that was working on the ash leaf cone roller problem (Evenden 2009). The Apanteles sp. was still the dominant parasitoid and so, along with studies on the chemical ecology of the moth (Wist et al. 2014), I also studied the third trophic level in this system (Wist and Evenden 2013). Of course, I couldn’t go through my studies without knowing what the species designation was for the dominant parasitoid wasp. Fortunately, Jose Fernandez-Triana had just begun his study of the genus Apanteles at the CNC in Ottawa and once Henri Goulet passed along the Apanteles specimens that I had sent for identification he quickly determined that this parasitoid was Apanteles polychrosidis Viereck (Hymenopetra: Braconidae) (Fig. 2).
Apanteles polychrosidis kills the ash leaf cone roller larvae before they can chew their emergence “window” that they use to escape the cone rolled leaflet as adults. This behaviour gives a fairly reliable visual cue that a cone rolled leaflet without a “window” has been parasitized by A. polychrosidis because the other parasitoids in the complex emerge after the cone roller has pupated and created its escape route “window”. Unrolling the leaflet confirms the presence of A. polychrosidis if its telltale “hammock-like” cocoon is present (Fig. 3). This type of cocoon is thought to be a defense against hyper-parasitism but as we found (Wist and Evenden 2013) it doesn’t always work out for A. polychrosidis!

Fig. 3 – Apanteles polychrosidis Viereck (Hymenopetra: Braconidae) adult above its cocoon and beside the leaflet cone rolled by Caloptilia fraxinella (Ely) (Lepidoptera: Gracillaridae). Note the emergence hole in the side of the leaflet that the wasp chewed to escape.
To assess the percentage of parasitism by this dominant parasitoid I adapted a method that Chris Saunders and I had discussed years earlier for assessing the parasitism of Apanteles sp. on individual trees. For the initial experiment in our paper (Wist et al. 2015) I sampled leaflets to estimate the density of cone rollers on the tree and estimated the percentage of parasitism by A. polychrosidis on two of the common urban species of ash in Edmonton. Apanteles polychrosidis parasitism was higher on black ash, F. nigra, at all sites than it was on green ash, F. pennsylvanica, which can be called differential parasitism and it seems to be common when host larvae develop on two or more host plants, but had not been well studied on trees. When host density and parasitism were graphed, the relationship of parasitism to host density could be visualized by the slope of the regression line, and on black ash, parasitism was independent of host density on black ash, but was negatively density dependent on green ash. In other words, on black ash parasitism is always high but on green ash, parasitism declines as the density of C. fraxinella increases. I ran the same experiment on green and black ash trees in Saskatoon with the same results but we chose to leave them out of the final version of the manuscript.
I was already studying the chemical ecology of C. fraxinella so this was where we looked for an answer to the differential parasitism in the field. I ran a y-tube olfactometer experiment with black and green ash plant material as the attractive source of volatile organic chemicals (VOCs) and this turned out to be rather tricky. I had three treatments that I wanted to test; undamaged leaflets, leaflets damaged by C. fraxinella and leaflets that were mechanically damaged.
First, I tried to bag small seedlings as the source of the plant smell but I couldn’t seal the system well enough to get reliable airflow through the y tube chamber. I had to switch to using leaflets alone which raises the issue of the smell of the leaflets changing once they have been removed from the tree which could be a problem especially in the “undamaged” treatment. I also needed enough female A. polychrosidis hunting for hosts to give me a decent sample size so I had to collect and emerge as many “un-windowed” cone-rolled leaflets as I could in the summer, and hope that they would actually mate and want to oviposit into host larvae at this point in their lives. Another issue was that I couldn’t coax my summer emerged C. fraxinella to lay eggs on ash seedlings to create leaf-mined treatments. Fortunately, a subset of the local population of C. fraxinella had developed a second generation on the new ash leaves that a dying ash tree puts out in July in an effort to save itself. These leaflets became my leaf-mined treatment. Over two seasons with a lot of juggling and timing of three species I was able to gather enough experimental data with the olfactometer to discover that female A. polychrosidis were differentially attracted to the volatile odour cues from each ash species. In green ash tests, they were attracted to the smell of green ash alone but in black ash tests, they were not attracted unless the leaflets were attacked by its host. The “icing on the manuscript cake” was the GC-EAD results by co-authour Regine Gries that showed that 13 compounds in the volatile profile of ash could be sensed by the antennae of A. polychrosidis, and some of them are known to increase in response to herbivore damage.
I’d say that this manuscript is a starting point for further studies on this interesting parasitism system and could accommodate projects from chemical ecology and landscape ecology perspectives at the very least. In fact, Danielle Hoefele and Sarah McPike have already begun projects in Maya’s lab on the Fraxinus–Caloptilia-Apanteles system. In case you’d like to know more, here is the link to our manuscript published in Arthropod-Plant Interactions.
This year’s 2015 Joint Annual Meeting in Montréal, Québec includes a free lunchtime workshop sponsored by Cambridge University Press that tackles the topic of publishing scientific papers.
Discussion will be led by a three-member panel examining the publication process through the eyes of an author (J. Saguez), a journal editor (K. Floate) and a publisher (D. Edwards). Following short presentations by each panelist, the floor will be opened for general questions and discussion.
Send us your questions and we will do our best to address them in our presentations.
What makes for a good paper? Who should I include as co-authors? How important is the cover letter? Why is the review process so long? How can I best respond to reviewer comments? What journal should I publish in? What is hybrid open access? What are predatory publishers? Why don’t journals make publications freely available? Knowing the answers to these and other questions can take some of the frustration out of the publication process.
Our goal is to ensure that everyone leaves with a full stomach and new insights to simplify the publication of their next paper! You can help us by sending your questions to Kevin Floate (Kevin.Floate@agr.gc.ca) by October 23rd.
See you in Montréal!
Julien Saguez – Independent Researcher/Author
Kevin Floate – Agriculture and Agri-Food Canada; Editor-in-Chief, The Canadian Entomologist
Daniel Edwards – Senior Commissioning Editor, Journals, Cambridge University Press
CONTACTER LA SOCIÉTÉ
Assistant administratif : info@esc-sec.ca
SEC Président : ESCPresident@esc-sec.ca
Follow The Society on Twitter
Tweets by @CanEntomologistThis post is also available in: English









































































